Autoclave Resistant Adhesives May Be the Answer
High heat, humidity, chemicals, and other environmental factors can cause challenges for medical tools like scopes, surgical instruments, dental equipment, general-use medical devices, and housing assemblies, as well as sensors and RFID tags adhered to medical tools. The materials that bond, coat, and encapsulate the components of such devices can be negatively impacted by these forces which can potentially lead to device failure, lost or misplaced instruments, or harm to a patient. That’s why it’s critical the products used to protect and bond the parts together be capable of tolerating a variety of stresses.
Along with the rigorous types of physical testing, standards, and approvals that medical device formulations are subjected to, including ISO 10993, many must also be able to withstand repeat cycles of sterilization.
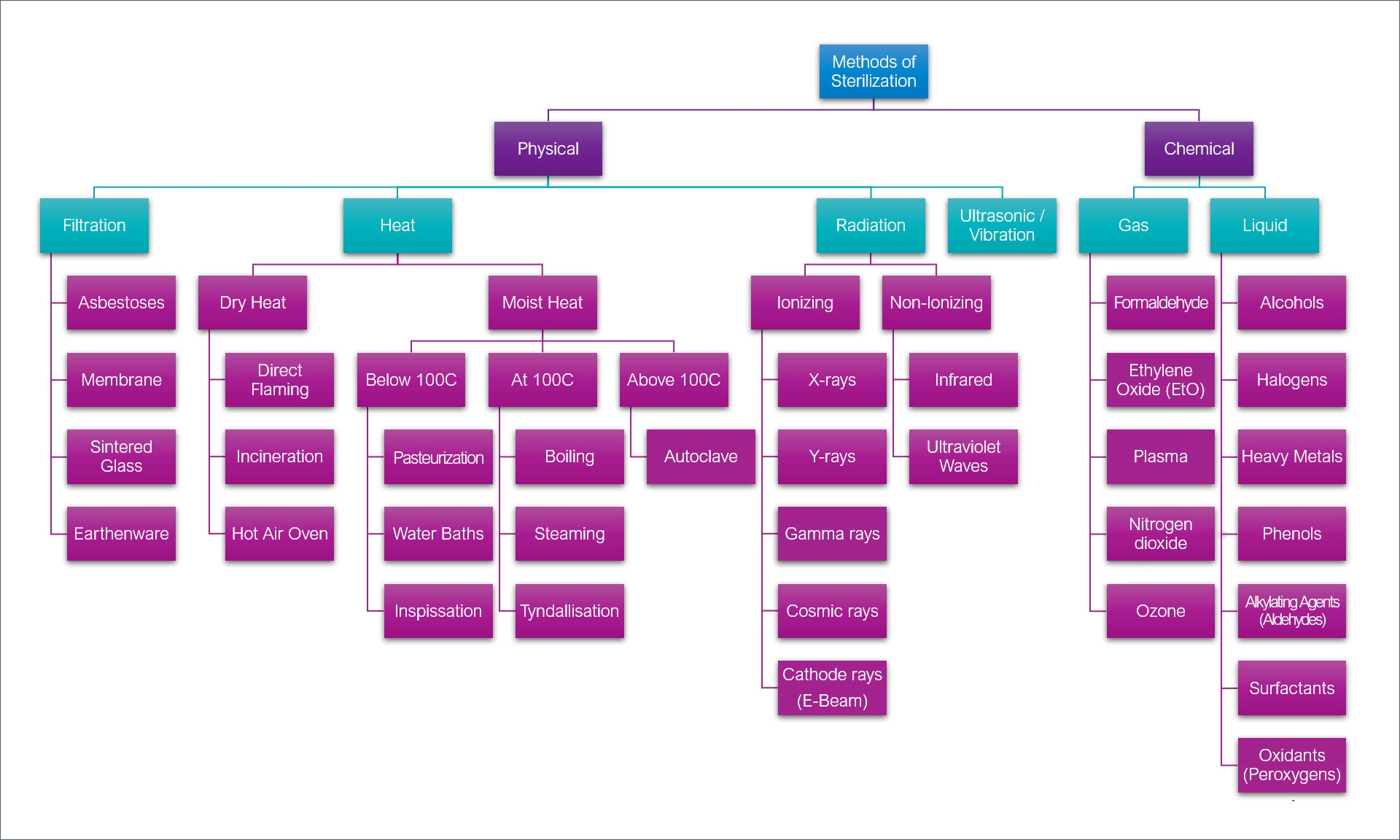
Two categories of sterilization used in the medical device industry are classified as either physical or chemical. Physical sterilization methods include filtration, heat, radiation, and ultrasonic/vibration, whereas chemical sterilization requires a gas or liquid process. There is a large subset of techniques that fall under these main classes, such as EtO, gamma ray, E-beam, cathode ray, and oxidants, as well as autoclave and STERRAD™ Hydrogen Peroxide-Plasma.
The types of physical and chemical sterilization methods that are commonly used throughout the medical industry.
There are pros and cons to using many of these processes. Positive attributes and outcomes depending on the method selected include high penetration capability, very rapid action against microorganisms, and the elimination of potential residues. Some of the negative factors can include long sterilization cycles, flammability, the use of hazardous materials or chemicals, yellowing of polymers, and corrosion of various metal substrates.
Considerations as to the kind of sterilization to employ are dependent upon the end-use product, its function, the substrate components, and the pressures and environments the device will be subjected to. Autoclave (steam) and Hydrogen Peroxide Gas Plasma (HPGP) are two methods often utilized in the medical device industry.
Autoclaving uses highly pressurized steam to sterilize instruments from microorganisms and bacteria that can potentially contaminate substrate surfaces by killing and rendering them ineffective. However, there are critical issues to keep in mind. Many metals have the potential to corrode or rust after repeated exposure to steam. Some plastics may lose their structural integrity, be sensitive to elevated levels of heat, be susceptible to migration of plasticizers to the substrate surface, or negatively react or break down when exposed to water.
Sterilization by autoclaving is limited to certain applications. Many medical device adhesives, for instance, have a difficult time holding up to autoclaving because of the high temperature, humidity, and pressure involved in the process. Due to these factors, there can be a weakened bond joint that fails. Alternatives to steam sterilization for heat and moisture-sensitive instruments and devices are being considered more regularly, although autoclave remains one of the most accessible options for hospitals, laboratories, research facilities, etc.
STERRAD™ Sterilization is an ideal method for heat and moisture-sensitive devices as temperatures do not exceed ~56°C (133°F) during processing. A solution of hydrogen peroxide and water is delivered to the sterilizer, then vaporized into the chamber. The solution surrounds and interacts with devices and creates a biocidal environment that inactivates microorganisms. A strong electrical field is applied to the chamber and creates a hydrogen peroxide gas plasma that dissociates hydrogen peroxide molecules into energized species. Once the electrical field is turned off, the energized species recombine, which turns the hydrogen peroxide into water and oxygen. This ensures the instruments are rapidly sterilized without any toxic residue remaining.
Many types of medical scopes and devices require repeat sterilization cycles or multiple instances of autoclaving or alternative sterilization methods like Hydrogen Peroxide-Plasma Sterilization.
Sample List of SCOPES Requiring Multiple Sterilization Cycles
Arthroscopes
Bronchoscopes
Colonoscopes
Duodenoscopes
Endoscopes
Esophagoscopes
Laparoscopic Instruments
Sample List of Additional DEVICES
Dental Tools/Equipment
Ophthalmic Tools/Equipment
Surgical Tools/Instruments
Endoscope Biopsy Accessories (typically tools, ex. forceps)
Irrigation Systems (ex., reusable irrigation cannulas)
One important application that has come to the forefront of the medical device industry is the use of identification tags, or RFIDs (radio frequency identification), on surgical tools, instruments, and packaging such as vials. RFIDs on medical implements aid in the maintenance of accurate inventory levels and help keep track of them during procedures. Other areas of use and typical applications include:
Hospitals, Clinics, and Caregivers:
- “Check in / check out” the multitude of devices and tools required for procedures and surgeries
− Driven by cases of tools or accessories being left within the body after surgery - Validate sterilization status before use
- Enhance inventory detail and data
Other Healthcare Areas:
- Pharmaceutical tracking to ensure proper dosing is provided and proper storage conditions are met
- Blood / biologic sample tracking for storage, use times, and automatic tracking to ensure proper handling
- Assets such as surgical tools, dental and hospital equipment, patients, and other
RFIDs that are affixed to often-used surgical tools and the materials used to adhere them, must pass multiple instances of sterilization and be successful in enduring these processes for hundreds of cycles.
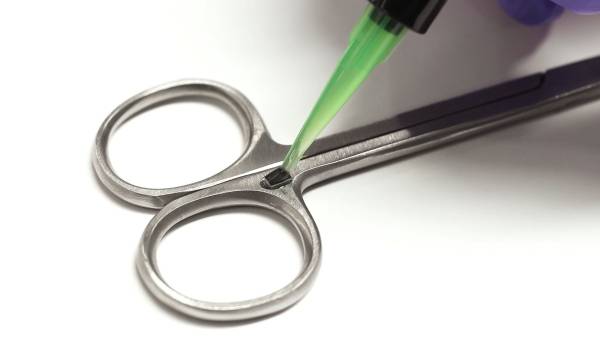
Application of encapsulant being applied over an RFID chip attached to medical tweezers.
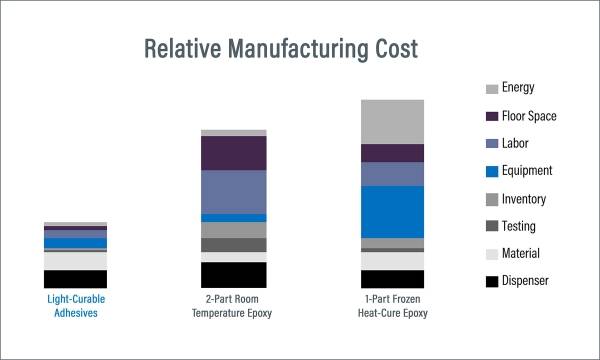
Although there are formulated one- and two-part epoxies on the market for bonding, coating, and encapsulating medical devices and RFIDs, these products may not be the best choice. When compared to light-curable materials, epoxies pose numerous disadvantages such as specialized mixing systems, long cure times of up to 60 minutes, short pot life, the use of heat-curing ovens (in some instances), lower flexibility and impact resistance, and the need for purge cycles which can result in hazardous waste. One-component light-curable adhesives provide an alternative to these solutions by offering manufacturers fast cure times from 1-30 seconds, no mixing or purging, a range of grades from rigid to flexible, RoHS-compliant, and no solvents added formulations for easier handling and disposal. The positive benefits of light-curable materials can help enhance manufacturing efficiency, decrease costs, and increase throughput.
To fulfill the need for a product capable of surviving frequent sterilization sequences, a new versatile UV/LED curable adhesive was developed that exhibits extremely low water absorption (0.5%) and is resistant to 100+ cycles of autoclave and plasma processes. This material is recommended for use as a coating or encapsulant for housing assemblies, single-use or multiple-use medical instruments, and a variety of medical scopes and dental equipment. It is especially suited for encapsulating RFID chips, sensors, and other electronic components found on medical devices, tools, and vials where moisture ingression may be of concern. Bondable substrates include stainless steel, aluminum, glass, PP/PE, and printed circuit boards.
The adhesive was subjected to a range of performance tests that included accelerated aging and sterilization, water soak evaluation, electrical property examination, and water vapor transmission rate testing. Compliant with ISO 10993-5 cytotoxicity standards, the material cures in seconds upon exposure to broad-spectrum UV or 365 nm LED light.
If you’re a manufacturer involved in the assembly and protection of medical devices and instruments and are seeking a bonding, coating, and encapsulating material that can withstand 100+ cycles of sterilization, then this product may be a solution for your application.
STERRAD™ is a registered trademark of ASP Global Manufacturing, GmbH.
_________________________________________________________
Enjoying This Content? Let’s Stay Connected.
If you’re finding value in our insights, why not get more of it—delivered right to your inbox? Subscribe to receive the latest technical articles, white papers, product news, and expert tips.